COVID-19 was first reported in Wuhan, China, at the end of December, 2019.1 The disease is an acute respiratory illness ranging in severity from mild to severe, with death in some cases; many infected people are asymptomatic. Since the end of January, 2020, cases of COVID-19 have been reported in more than 200 countries around the world. On March 11, 2020, WHO described the spread of COVID-19 as a pandemic.2
The causative agent of COVID-19 is the betacoronavirus severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). SARS-CoV-2 can be transmitted in many ways, with the main route of transmission via contact with infected people (eg, by secretions, particularly droplets).3 As of Aug 15, 2020, there have been more than 21 million laboratory-confirmed cases of SARS-CoV-2 infection, and more than 750 000 deaths.1
Because of the rapid global spread of SARS-CoV-2 infection and the high mortality rate, development of a vaccine is an urgent task. Vaccination will restrict the spread of COVID-19 and reduce mortality. Intensive research and development of vaccines is currently underway in China, Russia, the UK, the USA, and other countries.4 According to WHO, on Aug 13, 2020, 29 candidate COVID-19 vaccines based on different platforms (vectored, DNA, mRNA, inactivated, etc) were being tested in clinical trials.4
Prevention of SARS-CoV-2 infection might be achieved by targeting the spike protein (glycoprotein S), which interacts with the ACE2 receptor and enables entry of SARS-CoV-2 into the cell. Blocking this interaction decreases viral internalisation and replication.5, 6, 7 Most vaccines that are currently in development target glycoprotein S as the main antigen. The structure and function of the SARS-CoV-2 glycoprotein S is similar to that of other highly pathogenic betacoronaviruses, such as Middle East respiratory syndrome coronavirus (MERS-CoV) and severe acute respiratory syndrome coronavirus (SARS-CoV).8 Glycoprotein S consists of two subunits: S1 contains a receptor-binding domain (RBD), which interacts with the ACE2 receptor on the cell surface; S2 mediates the fusion of viral and cell membranes via formation of a six-helix bundle fusion core.9, 10 To protect against SARS-CoV-2 infection, it is important to form neutralising antibodies targeting S1 RBD, S1 N-terminal domain, or the S2 region; these antibodies block binding of the RBD to the ACE2 receptor and prevent S2-mediated membrane fusion or entry into the host cell, thus inhibiting viral infection.11, 12
When developing a vaccine (particularly during a pandemic), it is important to consider that a protective response must develop in a short time (eg, up to 1 month). Moreover, previous work on vaccines for MERS-CoV13 and SARS-CoV14 showed that both humoral and cellular (cytotoxic) immune responses are important to induce a protective immune response. To achieve these goals, one of the most attractive options is for vaccines to be based on recombinant viral vectors, which can induce humoral and cellular immune responses and form protective immunity after one or two doses.15, 16 Recombinant adenovirus vectors have been used for a long time, with safety confirmed in many clinical studies of various preventive and therapeutic drugs.17, 18, 19, 20, 21, 22, 23 Moreover, the long-term effects of vectors based on adenoviruses have been investigated,23 by contrast with newer methods that remain to be studied long term. For formation of a robust long-lasting immune response, a prime-boost vaccination is advisable, which is widely used with registered vaccines for diseases including hepatitis B24 and Ebola virus disease.25 When using vector-based vaccines, immune responses are formed not only to the target antigen but also to the vector component. As a result, the best vaccination scheme is heterologous vaccination, when different viral vectors are used to overcome any negative effects of immune response to vector components.25, 26, 27 Such an approach was successfully used with an Ebola virus disease vaccine developed in Russia and licensed in 2015.25
We designed a novel, heterologous adenoviral vector-based vaccine against SARS-CoV-2 suitable for prime-boost vaccination. The vaccine was designed with two recombinant adenovirus vectors and was developed as two formulations (frozen [Gam-COVID-Vac] and lyophilised [Gam-COVID-Vac-Lyo]). We aimed to assess safety and immunogenicity of both vaccine formulations and to compare the humoral immune response with that recorded in people who have recovered from COVID-19.
Between June 18 and Aug 3, 2020, 76 healthy adults were enrolled to the two studies from the volunteer register (figure 1). 43 adults were selected at the beginning of each study from the volunteer registry; 38 participants were included in each study and five people were kept as bac
 Red AlbanteSep 05, 2020 03:29(Article#20) Keep posting! Follow me on Instagram/Twitter: Redalbante
Red AlbanteSep 05, 2020 03:29(Article#20) Keep posting! Follow me on Instagram/Twitter: Redalbante Joyce Sta. AnaSep 05, 2020 12:44
Joyce Sta. AnaSep 05, 2020 12:44 " target="_blank">https://www.goodinfonet.com/img/emoticons/saddy.png'>
" target="_blank">https://www.goodinfonet.com/img/emoticons/saddy.png'>
 BJ_ JoseSep 05, 2020 11:38Hope so
BJ_ JoseSep 05, 2020 11:38Hope so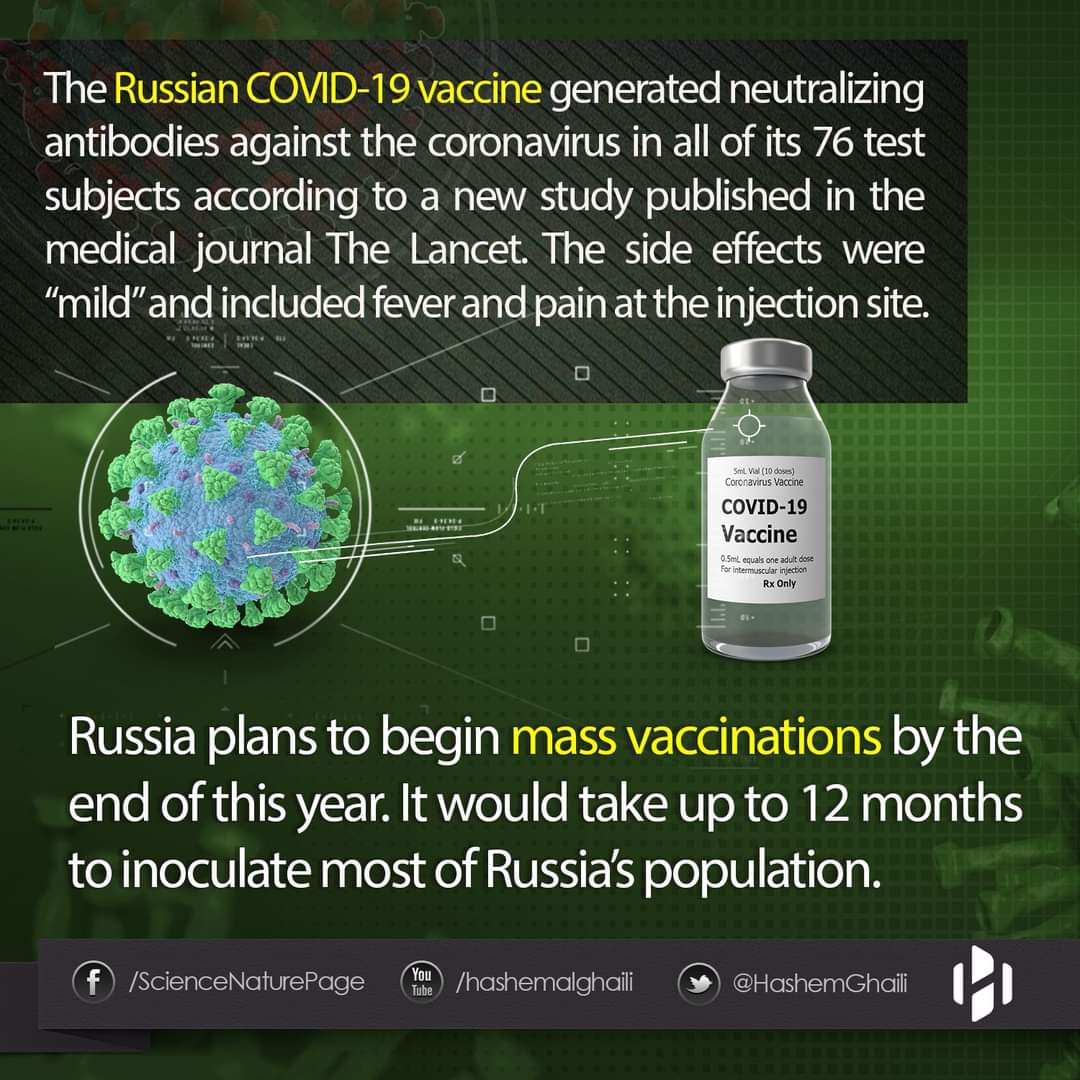










.jpeg)








Comments